Chapter 2: Q4.69P (page 40)
Give the oxidation number of phosphorus in the following:
Short Answer
You need to provide the oxidation no of phosphorus for the given compounds.
Learning Materials
Features
Discover
Chapter 2: Q4.69P (page 40)
Give the oxidation number of phosphorus in the following:
You need to provide the oxidation no of phosphorus for the given compounds.
All the tools & learning materials you need for study success - in one app.
Get started for free
Show, with calculations, how the following data illustrate the law of multiple proportions:Compound 1: 77.6 mass % xenon and 22.4 mass % fluorineCompound 2: 63.3 mass % xenon and 36.7 mass % fluorine
Correct the formula to match the name of the following compounds:
Each scene below represents a mixture. Describe each one in terms of the number of elements and/or compounds present.


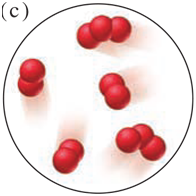
Each scene below represents a mixture. Describe each one in terms of the number of elements and/or compounds present.
Explain the following statement: The smallest particles unique to a compound cannot be atoms.
What do you think about this solution?
We value your feedback to improve our textbook solutions.