Chapter 2: The Components of Matter
Q15.95 P
Draw the R group of (a) glycine; (b) isoleucine; (c) tyrosine.
Q15.98 P
Write the sequence of the complementary DNA strand that pairs with each of the following DNA base sequences:
(a) TTAGCC
(b) AGACAT
Q15.99 P
Question: Write the sequence of the complementary DNA strand that pairs with each of the following DNA base sequences:
(a) GGTTAC
(b) CCCGAA
Q15P
Which of the following scenes illustrate(s) the fact that compounds of chlorine (green) and oxygen (red) exhibit the law of multiple proportions? Name the compounds.
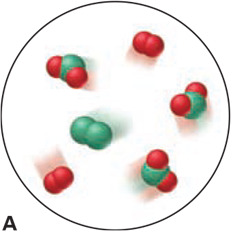
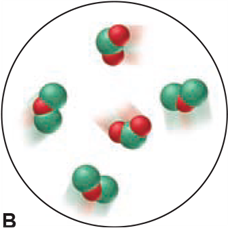

Q160CP
Liquid ammonia auto ionizes like water:
where (am) represents solvation by.
(a) Write the ion-product constant expression, Kam.
b) What are the strongest acid and base that can exist in(l)?
(c)and HCOOH are levelled in(l). Explain with equations.
(d) At the boiling point(),. Calculate [] at this temperature.
(e) Pure sulfuric acid also auto ionizes. Write the ion-product constant expression,, and find the concentration of the conjugate base at
Q162CP
Thiamine hydrochloride is a water soluble form of thiamine (vitamin B1; . How many grams of the hydrochloride must be dissolved in 10.00mL of water to give a pH of 3.50?
Q167CP
At and 1 atm,. At these conditions, find
(a)in pure water,
(b)in 0.010 MNaOH , and
(c) [OH] in.
(d) Findin 0.0100 MKOH atand 1000atm (Kw = 1.101012).
(e) Find the pH of pure water atand 1000atm
Q16P
(a) Does the percent by mass of each element in a compound depend on the amount of compound? Explain.
(b) Does the mass of each element in a compound depend on the amount of compound? Explain.
Q171CP
The Group 5A(15)hydrides react with boron trihalides in a reversible Lewis acid-base reaction. When 0.15mol of (s) is introduced into a3.0 -Container at a certain temperature,mol ofis present at equilibrium:
(a) Findfor the reaction at this temperature.
(b) Draw a Lewis structure for the reactant
Q172CP
A 1000m solution of chloroacetic acidfreezes at . Find the Ka of chloroacetic acid. (Assume the molarities equal the molalities.)
