Chapter 2: Q15.95 P (page 40)
Draw the R group of (a) glycine; (b) isoleucine; (c) tyrosine.
Short Answer
- Glycine's side chain is H.
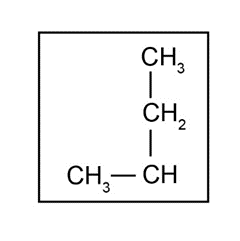
- The isoleucine side chain is,
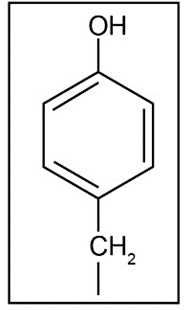
c. Tyrosine's side chain is,
Learning Materials
Features
Discover
Chapter 2: Q15.95 P (page 40)
Draw the R group of (a) glycine; (b) isoleucine; (c) tyrosine.

c. Tyrosine's side chain is,

All the tools & learning materials you need for study success - in one app.
Get started for free
Even though several elements have only one naturally occurring isotope and atomic nuclei have whole numbers of protons and neutrons, no atomic mass is a whole number. Use the data from Table 2.2 to explain this fact.
Choose the correct answer. The difference between the mass number of an isotope and its atomic number is (a)directly related to the identity of the element; (b) the number of electrons; (c) the number of neutrons; (d) the number of isotopes.
In our modern view of matter and energy, is the law of mass conservation still relevant to chemical reactions? Explain.
What is the key difference between an element and a compound?
Give the name, atomic symbol, and group number of the element with the following Z value, and classify it as a metal, metalloid, or nonmetal:
(a) Z = 33 (b) Z = 20 (c) Z = 35 (d) Z = 19 (e) Z = 13
What do you think about this solution?
We value your feedback to improve our textbook solutions.