Chapter 14: Q82P (page 622)
How do atomic properties account for the enormous number of carbon compounds? Why don’t other group 4A(14) elements behave similarly?
Short Answer
Due to the tetravalent nature of carbon.
Learning Materials
Features
Discover
Chapter 14: Q82P (page 622)
How do atomic properties account for the enormous number of carbon compounds? Why don’t other group 4A(14) elements behave similarly?
Due to the tetravalent nature of carbon.
All the tools & learning materials you need for study success - in one app.
Get started for free
Thallium forms the compound . What is the apparent oxidation state of Tl in this compound? Given that the anion is , what is the actual oxidation state of Tl? Draw the shape of the anion, giving its VSEPR class and bond angles. Propose a reason why the compound does not exist as.
The interhalogen IF undergoes the reaction depicted below (I is purple and F is green):
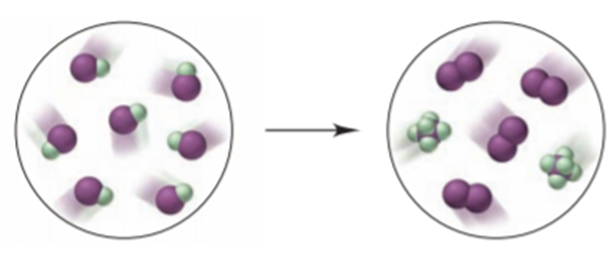
(a) Write the balanced equation.
(b) Name the interhalogen product.
(c) What type of reaction is shown?
(d) If each molecule of IF represents mol, what mass of each product forms?
Question: As you move down Group 5A (15), the melting points of the elements increases and then decreases. Explain
Question: Complete and balance the following:
From its formula, one might expect CO to be quite polar, but its dipole moment is low (0.11 D).
(a) Draw the Lewis structure for CO.
(b) Calculate the formal charges.
(c) Based on your answers to parts (a) and (b), explain why the dipole moment is so low
What do you think about this solution?
We value your feedback to improve our textbook solutions.