Chapter 15: Q15.7P (page 678)
Which of these bonds to carbon would you expect to be relatively reactive: ? Explain.
Short Answer
The answer is,
C=O
Learning Materials
Features
Discover
Chapter 15: Q15.7P (page 678)
Which of these bonds to carbon would you expect to be relatively reactive: ? Explain.
The answer is,
C=O
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw correct structures, by making a single change for any that are incorrect:
(a) 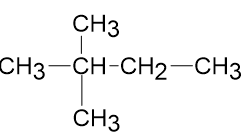
(b)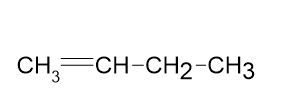
(c)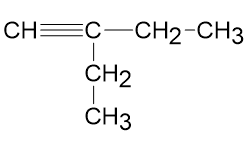
(d)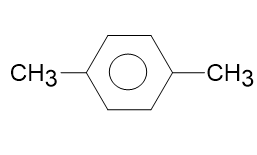
Define each type of isomer: (a) constitutional; (b) geometric; (c) optical. Which types of isomers are stereoisomers?
Among alkenes, alkynes and aromatic hydrocarbons, only alkenes exhibit cis-trans isomerism. Why don’t the others?
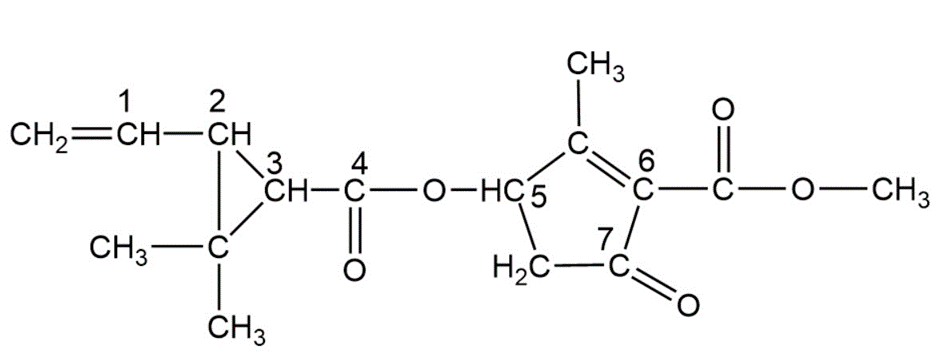
Pyrethrins, such as jasmolin II (below), are a group of natural compounds synthesised by flowers of the genus Chrysanthemum (known as pyrethrum flowers) to act as insecticides.
(a) Circle and name the functional groups in jasmolin II.
(b) What is the hybridization of the numbered carbons?
(c) Which, if any, of the numbered carbons are chiral centers?
Question: Which features of the retinal make it so valuable as a photon absorber in the visual systems of organisms?
What do you think about this solution?
We value your feedback to improve our textbook solutions.