Some of the most useful compounds for organic synthesis are Grignard reagents (general formula , where X is a halogen), which are made by combining an alkyl halide, , with. They are used to change the carbon skeleton of a starting carbonyl compound in a reaction similar to that with :
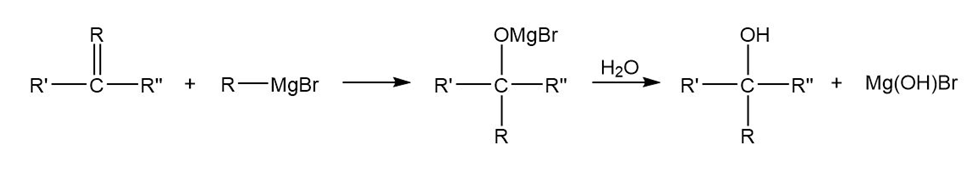
(a) What is the product, after a final step with water, of the reaction between ethanal and the Grignard reagent of bromobenzene?
(b) What is the product, after a final step with water, of the reaction between 2-butanone and the Grignard reagent of 2-bromopropane?
(c) There are often two (or more) combinations of Grignard reagent and carbonyl compound that will give the same product. Choose another pair of reactants to give the product in (a).
(d) What ±OH group at the end of the carbon chain?
(e) What Grignard carbonyl compound must react with a Grignard reagent to yield a product with the reagent and carbonyl compound would you use to prepare 2-methyl-2-butanol?







