Chapter 24: Q8 P (page 1103)
Write balanced nuclear equations for the following:
(a) Alpha decay of
(b) Electron capture by neptunium-232
(c) Positron emission by
Short Answer
The required balanced equations are
Learning Materials
Features
Discover
Chapter 24: Q8 P (page 1103)
Write balanced nuclear equations for the following:
(a) Alpha decay of
(b) Electron capture by neptunium-232
(c) Positron emission by
The required balanced equations are
All the tools & learning materials you need for study success - in one app.
Get started for free
If of the atoms of a radioactive isotope disintegrate in , what is the decay constant of the process?
Which isotope in each pair is more stable? Why?
(a) or (b)or
(c) or (d) or
What is the binding energy per nucleon? Why is the binding energy per nucleon, rather than per nuclide, used to compare nuclide stability?
Plutonium- represents a serious nuclear waste hazard. If seven half-lives are required to reach a tolerable level of radioactivity, how long must be stored?
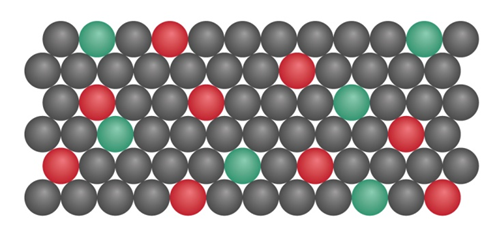
begins a decay series that ultimately forms 206Pb . The scene below depicts the relative number of atoms (red) and atoms (green) in a mineral. If all the Pb comes from , calculate the age of the sample.
What do you think about this solution?
We value your feedback to improve our textbook solutions.