Chapter 19: Q19.74P (page 875)
Find the molar solubility of in
(a) pure water and
(b) localid="1663327059323" .
Short Answer
(a) The molar solubility of in pure water is .
(b) The molar solubility of in is .
Learning Materials
Features
Discover
Chapter 19: Q19.74P (page 875)
Find the molar solubility of in
(a) pure water and
(b) localid="1663327059323" .
(a) The molar solubility of in pure water is .
(b) The molar solubility of in is .
All the tools & learning materials you need for study success - in one app.
Get started for free
solution contains and. Whatdata-custom-editor="chemistry" is required to precipitate the maximum amount ofbut none of the? (See Appendix C.)
An eco-botanist separates the components of a tropical bark extract by chromatography. She discovers a large proportion of quinidine, a dextrorotatory isomer of quinine used for control of arrhythmic heartbeat. Quinidine has two basic nitrogen’s and ). To measure the concentration, she carries out a titration. Because of the low solubility of quinidine, she first protonates both nitrogen’s with excessand titrates the acidified solution with standardized base. A -mg sample of quinidine is acidified withof.
(a) How many milliliters ofare needed to titrate the excess?
(b) How many additional milliliters of titrant are needed to reach the first equivalence point of quinidine dihydrochloride?
(c) What is theat the first equivalence point?
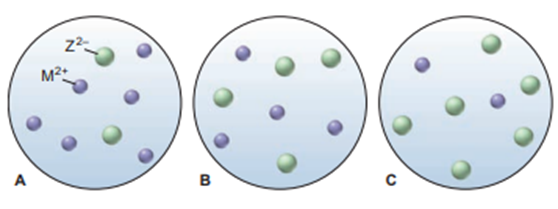
Scenes A to C represent aqueous solutions of the slightly soluble salt MZ (only the ions of this salt are shown):
(a) Which scene represents the solution just after solid MZ is stirred thoroughly in distilled water?
(b) If each sphere represents of ions, what is the of MZ?
(c) Which scene represents the solution afteris added?
(d) If is, which scene represents the solution after thehas been lowered?
Choose specific acid-base conjugate pairs to make the following buffers: (a) ; (b)
. (See Appendix C.)
Why does the color change of an indicator take place over a range of about 2 pH units?
What do you think about this solution?
We value your feedback to improve our textbook solutions.