Chapter 19: Q19.154 CP (page 878)
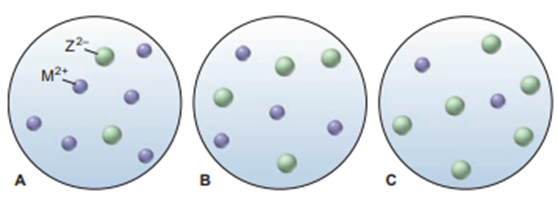
Scenes A to C represent aqueous solutions of the slightly soluble salt MZ (only the ions of this salt are shown):
(a) Which scene represents the solution just after solid MZ is stirred thoroughly in distilled water?
(b) If each sphere represents of ions, what is the of MZ?
(c) Which scene represents the solution afteris added?
(d) If is, which scene represents the solution after thehas been lowered?
Short Answer
- The scene represents the solution just after solid MZ is stirred thoroughly in distilled water is picture B.
- The value of MZ is .
- The picture C is the correct situation.
- The correct option is picture A.


