Chapter 19: Q19.69P (page 875)
Write the ion-product expressions for
Short Answer
The ion-product expression of the compounds is,
Learning Materials
Features
Discover
Chapter 19: Q19.69P (page 875)
Write the ion-product expressions for
The ion-product expression of the compounds is,
All the tools & learning materials you need for study success - in one app.
Get started for free
student wants to dissolve the maximum amount of to make of aqueous solution.
(a) Into which of the following solvents should she dissolve the salt?
localid="1664178101553"
(III) (IV) (V)
(b) Which would dissolve the least amount of salt?
What is when25.0 mL each of 0.044 M and 0.57 M are mixed[of ]?
A buffer that containsandhas a pH of . What is the pHaftermol of HCLis added toof this solution?
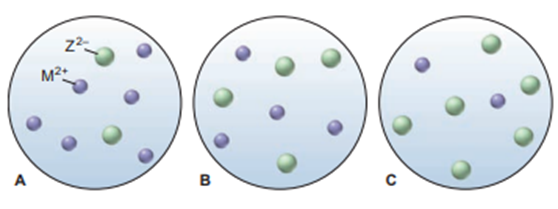
Scenes A to C represent aqueous solutions of the slightly soluble salt MZ (only the ions of this salt are shown):
(a) Which scene represents the solution just after solid MZ is stirred thoroughly in distilled water?
(b) If each sphere represents of ions, what is the of MZ?
(c) Which scene represents the solution afteris added?
(d) If is, which scene represents the solution after thehas been lowered?
What species are in the buffer region of a weak acid–strong base titration? How are they different from the species at the equivalence point? How are they different from the species in the buffer region of a weak base–strong acid titration?
What do you think about this solution?
We value your feedback to improve our textbook solutions.