Chapter 5: Q5.10P (page 227)
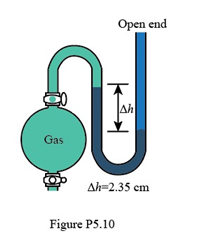
In Figure P5.10, what is the pressure of the gas in the flask (in atm) if the barometer reads 738.5 torr?
Short Answer
The pressure of the gas is 0.941 atm.
Learning Materials
Features
Discover
Chapter 5: Q5.10P (page 227)
In Figure P5.10, what is the pressure of the gas in the flask (in atm) if the barometer reads 738.5 torr?

The pressure of the gas is 0.941 atm.
All the tools & learning materials you need for study success - in one app.
Get started for free
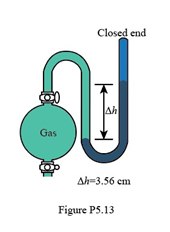
What is the pressure (in Pa) of the gas in the flask in Figure P5.13?
A 75.0-g sample of dinitrogen monoxide is confined in a 3.1-L vessel. What is the pressure (in atm) at ?
You have 357 mL of chlorine trifluoride gas at 699 mmHg and . What is the mass (in g) of the sample?
Are the particles in a gas farther apart or closer together than the particles in a liquid? Use your answer to explain each of the following general observations:
(a) Gases are more compressible than liquids.
(b) Gases have lower viscosities than liquids.
(c) After thorough stirring, all gas mixtures are solutions.
(d) The density of a substance in the gas state is lower than in the liquid state.
In a closed-end manometer, the mercury level in the arm attached to the flask can never be higher than the mercury level in the other arm, whereas in an open-end manometer, it can be higher. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.