Chapter 21: Q21.96P (page 922)
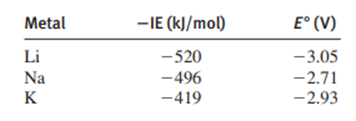
Identify those elements that can be prepared by electrolysis of their aqueous salts: fluorine, manganese, iron, cadmium.
Short Answer
Cadmium, Manganese, and Iron are the elements that can be prepared by electrolysis of their aqueous salts.