Chapter 21: Q21.62P (page 922)
Calculatefor each of the reactions
Short Answer
a.
b.
Learning Materials
Features
Discover
Chapter 21: Q21.62P (page 922)
Calculatefor each of the reactions
a.
b.
All the tools & learning materials you need for study success - in one app.
Get started for free
When a piece of metal A is placed in a solution containing ions of metal , metal plates out on the piece of A.
(a) Which metal is being oxidized?
(b) Which metal is being displaced?
(c) Which metal would you use as the anode in a voltaic cell incorporating these two metals?
(d) If bubbles of form when is placed in acid, will they form if A is placed in acid? Explain
Use Appendix D to calculate theof AgCl.
Comparing the standard electrode potentials of the Group metalswith the negative of their first ionization energies reveals a discrepancy:
Ionization process reversed:
Electrode reaction:
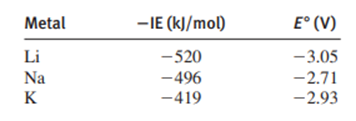
Note that the electrode potentials do not decrease smoothly down the group, as the ionization energies do. You might expect that if it is more difficult to remove an electron from an atom to form a gaseous ion (larger), then it would be less difficult to add an electron to an aqueous ion to form an atom (smaller), yetis more difficult to reduce than. Applying Hess’s law, use an approach similar to that for a Born-Haber cycle to break down the process occurring at the electrode into three steps and label the energy involved in each step. How can you account for the discrepancy?
Use the emf series (Appendix D) to arrange the species.
(a) In order of decreasing strength as oxidizing agents:
(b) In order of increasing strength as oxidizingagents :,
Why must an electrochemical process involve a redox reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.