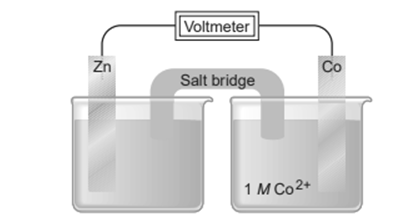
Chapter 21: Q21.40P (page 974)
Use the emf series (Appendix D) to arrange the species.
(a) In order of decreasing strength as oxidizing agents:
(b) In order of increasing strength as oxidizingagents :,
Short Answer
The required work use the emf series increasing and decreasing order strength as oxidizing agents given the highest and smallest value.