Chapter 21: Q21.22 P (page 973)
Consider the following general voltaic cell:
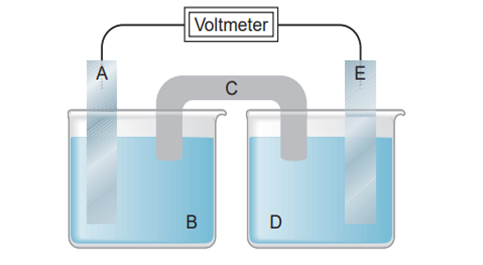
Identify the (a) anode, (b) cathode, (c) salt bridge, (d) electrode at which leave the cell, (e) electrode with a positive charge, and electrode that gains mass as the cell operates (assuming that a metal plates out).
Short Answer
(a) A
(b) E
(c) C
(d) A
(e) E
(f) E

