Chapter 18: Q18.65P (page 825)
Nitrous acid,, has aof. What areandin?
Short Answer
The values for and are –
Learning Materials
Features
Discover
Chapter 18: Q18.65P (page 825)
Nitrous acid,, has aof. What areandin?
The values for and are –
All the tools & learning materials you need for study success - in one app.
Get started for free
The beakers below depict the aqueous dissociations of weak acids HA (blue and green) and HB (blue and yellow); solvent molecules are omitted for clarity. If the HA solution is0.50L, and the HB solution is 0.25L, and each particle representsmol, find the Ka of each acid. Which acid, if either, is stronger?

Question: Use Appendix C to rank the following in order of increasing acid strength: .
Explain the differing extents of dissociation of in and .
Many molecules with central atoms from Period 3 or higher take part in Lewis acid-base reactions in which the central atom expands its valence shell. SnCl4 reacts with (CH3)3N as follows:
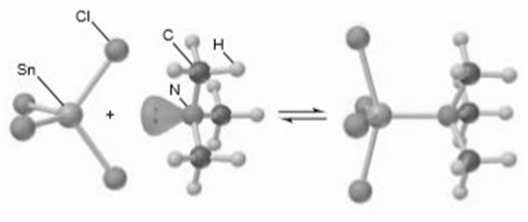
(a) Identify the Lewis acid and the Lewis base in the reaction.
(b) Give the nl designation of the sublevel of the central atom in the acid before it accepts the lone pair.
When salts are dissolved in water, the solution becomes acidic due to formation ofand.The overall process involves both Lewis and Brønsted-Lowry acid-base reactions. Write the equations for the process.
What do you think about this solution?
We value your feedback to improve our textbook solutions.