Chapter 18: Q18.120P (page 827)
Explain with equations and calculations, when necessary, whether an aqueous solution of each of these salts is acidic, basic, or neutral:
Short Answer
Answer
(a) Acidic
(b) Basic
(c) Acidic
Learning Materials
Features
Discover
Chapter 18: Q18.120P (page 827)
Explain with equations and calculations, when necessary, whether an aqueous solution of each of these salts is acidic, basic, or neutral:
Answer
(a) Acidic
(b) Basic
(c) Acidic
All the tools & learning materials you need for study success - in one app.
Get started for free
Write theexpression for each of the following in water:
(a)
(b)role="math" localid="1657022192179"
(c)role="math" localid="1657022265078"
Hypochlorous acid, , has a of . What are , , , and in ?
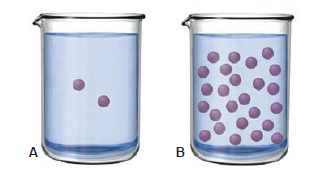
The two molecular scenes below depict the relative concentrations of (purple) in solutions of the same volume (with counter ions and solvent molecules omitted for clarity). If the pH in scene A is 4.8, what is the pH in scene B?
Polymers are not very soluble in water, but their solubility increases if they have charged groups.
(a) Casein, a milk protein, contains manygroups on its side chains. How does the solubility of casein vary with pH?
(b) Histones are proteins essential to the function of DNA. They are weakly basic due to the presence of side chains withand =NH groups. How does the solubility of a histone vary with pH?
(a) A Brønsted-Lowry acid-base reaction proceeds in the net direction in which a stronger acid and stronger base form a weaker acid and weaker base. Explain
(b) The molecular scene at the right depicts an aqueous solution of two conjugate acid-base pairs: HA/A and HB/B. The base in the first pair is represented by red spheres and the base in the second pair by green spheres; solvent molecules are omitted for clarity. Which is the stronger acid? Stronger base? Explain.

What do you think about this solution?
We value your feedback to improve our textbook solutions.