Chapter 18: Q165CP (page 829)
What is the pH of a vinegar with5.0 % (w/v) acetic acid in water?
Short Answer
The pH of the vinegar is 2.41.
Learning Materials
Features
Discover
Chapter 18: Q165CP (page 829)
What is the pH of a vinegar with5.0 % (w/v) acetic acid in water?
The pH of the vinegar is 2.41.
All the tools & learning materials you need for study success - in one app.
Get started for free
The following scenes represent three weak acids HA
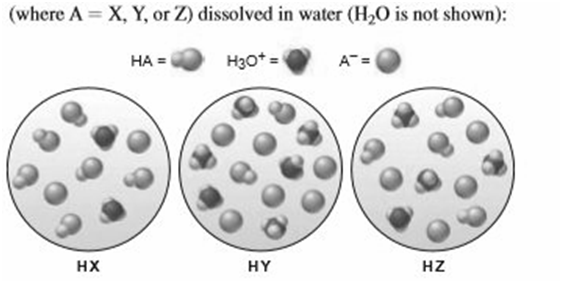
(a) Rank the acids in order of increasing Ka.
(b) Rank the acids in order of increasing
c) Rank the conjugate bases in order of increasing .
(d) What is the percent dissociation of HX?
(e) If equimolar amounts of the sodium salts of the acids (NaX, NaY, and NaZ) were dissolved in water, which solution would have the highest pOH? The lowest pH?
What is the difference between and for the autoionization of water?
Seashells are mostly calcium carbonate, which reacts with according to the equation
If increases at higher pressure, will seashells dissolve more rapidly near the surface of the ocean or at great depths? Explain.
Drinking water is often disinfected with , which hydrolyzes to form HClO, a weak acid but powerful disinfectant:
The fraction of HClO in solution is defined as
(a) What is the fraction of HClO at pH 7.00(Ka of HClO=?
(b) What is the fraction at pH 10.00?
The beakers shown contain 0.300 Lof aqueous solutions of a moderately weak acid HY. Each particle represents 0.010 mol; solvent molecules are omitted for clarity.
(a) The reaction in beaker Ais at equilibrium. Calculate Q for B, C, and D to determine which, if any, is also at equilibrium.
(b) For any not at equilibrium, in which direction does the reaction proceed?
(c) Does dilution affect the extent of dissociation of a weak acid? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.