Chapter 9: Q. 9.95 (page 318)
How many milliliters of a propyl alcohol solution would you need to obtain of propyl alcohol?
Short Answer
As an outcome, of liquid is forced to hold of solute.
Learning Materials
Features
Discover
Chapter 9: Q. 9.95 (page 318)
How many milliliters of a propyl alcohol solution would you need to obtain of propyl alcohol?
As an outcome, of liquid is forced to hold of solute.
All the tools & learning materials you need for study success - in one app.
Get started for free
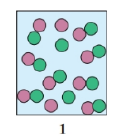
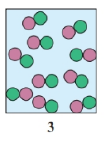
Select the diagram that represents the solution formed by a solute that is a
a. nonelectrolyte
b. weak electrolyte
c. strong electrolyte

What is the final volume, in milliliters, when . of each of the following solutions is diluted to provide the given concentration?
a. solution to give a solution
b. solution to give a solution
c. solution to give a solution
Indicate whether each of the following ionic compounds is soluble or insoluble in water:
a.
b.
c.
d.
e.
An intravenous solution contains of and of. Ifis the only cation in the solution, what is theconcentration, in milliequivalents per liter?
State whether each of the following refers to a saturated or an unsaturated solution:
a. A spoonful of salt added to boiling water dissolves.
b. A layer of sugar forms on the bottom of a glass of tea as ice is added.
c. A kidney stone of calcium phosphate forms in the kidneys when urine becomes concentrated.
What do you think about this solution?
We value your feedback to improve our textbook solutions.