Chapter 9: Q. 9.19 (page 294)
An intravenous solution contains of and of. Ifis the only cation in the solution, what is theconcentration, in milliequivalents per liter?
Short Answer
Concentration offor the solution is
Learning Materials
Features
Discover
Chapter 9: Q. 9.19 (page 294)
An intravenous solution contains of and of. Ifis the only cation in the solution, what is theconcentration, in milliequivalents per liter?
Concentration offor the solution is
All the tools & learning materials you need for study success - in one app.
Get started for free
Water is a polar solvent and hexane is a nonpolar solvent. In which solvent is each of the following, which is found or used in the body, more likely to be soluble?
a. vegetable oil, nonpolar
b. oleic acid (lipid), nonpolar
c. niacin (vitamin ), polar
d. (iron supplement), ionic
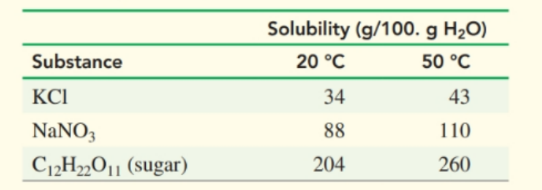
Determine whether each of the following solutions will be saturated or unsaturated at :
a. adding of to role="math" localid="1652588032291"
b. adding to role="math" localid="1652588024461"
c. adding of sugar to
State whether each of the following refers to a saturated or an unsaturated solution:
a. A spoonful of salt added to boiling water dissolves.
b. A layer of sugar forms on the bottom of a glass of tea as ice is added.
c. A kidney stone of calcium phosphate forms in the kidneys when urine becomes concentrated.
Potassium nitrate has a solubility of . Determine if each of the following forms an unsaturated or saturated solution at
a. adding localid="1653998424495"
b. adding localid="1653998430681"
c. addinglocalid="1653998451738"
Identify each of the following as characteristic of a solution, colloid, or suspension:
a. a mixture that cannot be separated by a semipermeable membrane
b. a mixture that settles out upon standing
What do you think about this solution?
We value your feedback to improve our textbook solutions.