Chapter 3: Q.3.68 (page 92)
Calculate the energy to heat two cubes (silver and copper), each with a volume of from .
Short Answer
the energy to heat two cubes (silver and copper) isandrespectively.
Learning Materials
Features
Discover
Chapter 3: Q.3.68 (page 92)
Calculate the energy to heat two cubes (silver and copper), each with a volume of from .
the energy to heat two cubes (silver and copper) isandrespectively.
All the tools & learning materials you need for study success - in one app.
Get started for free
A high-protein diet contains of carbohydrate, of fat, and of protein. How much energy, in kilocalories and kilojoules, does this diet provide? (Round off the answer for each food type to the tens place.)
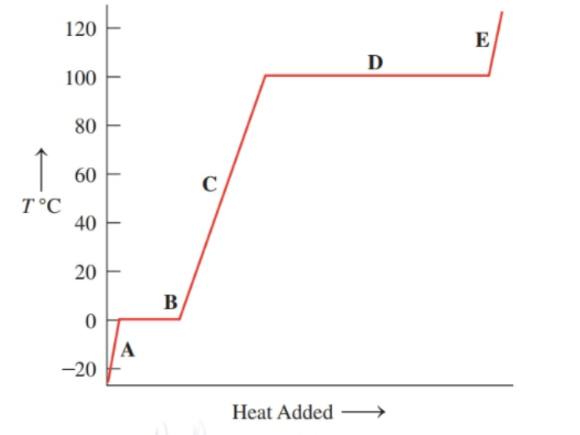
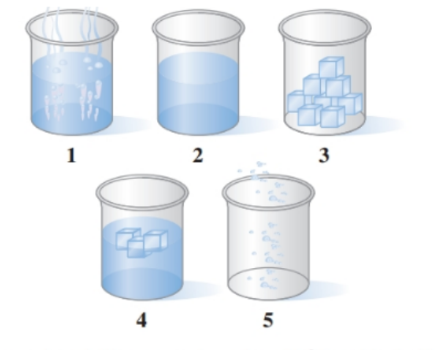
Associate the contents of the beakers ( to ) with segments (to ) on the following heating curve for water: ()
Classify each of the following mixtures as homogeneous of heterogeneous:
a. hot fudge sundae
b. herbal tea
c. vegetable oil
Classify each of the following pure substances as an element or a compound:
a. silicon () chip
b. hydrogen peroxide localid="1651737397761"
c. oxygen gas localid="1651737412373" role="math"
d. rust localid="1651737418279"
e. methane localid="1651737422197" in natural gas
Calculate the unknown temperature in each of the following:
a.
b.
c. localid="1651679546288"
d.
e.
What do you think about this solution?
We value your feedback to improve our textbook solutions.