Chapter 3: Q. 3.86 (page 93)
A sample of rice is placed in a calorimeter. When the sample is burned, is given off. What is the energy value for the rice?
Short Answer
The energy value of rice is burning a sample in a calorimeter.
Learning Materials
Features
Discover
Chapter 3: Q. 3.86 (page 93)
A sample of rice is placed in a calorimeter. When the sample is burned, is given off. What is the energy value for the rice?
The energy value of rice is burning a sample in a calorimeter.
All the tools & learning materials you need for study success - in one app.
Get started for free
Use the heat eyuatson to calculate the energy, in joules and calories, for each of the following (see Table 3.11):
a. to beatof water from to
b. to beatof copper from to
c. lost whenof ethanol, , cools from to
d. lost whenof iron cools from to
The highest recorded body temperature that a person has survived is . Calculate that temperature in degrees Fahrenheit and in kelvins.
Substances A and B are the same mass and at the same initial temperature. When they are boated, the final terafeature of is higher than the temperature of . What does this iell you about the specific heats of and ?
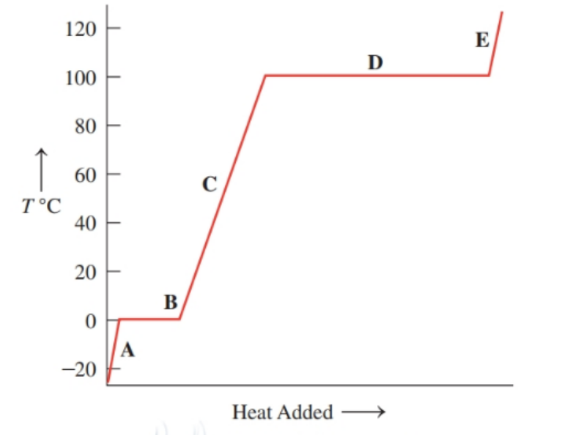
Associate the contents of the beakers ( to ) with segments (to ) on the following heating curve for water: ()
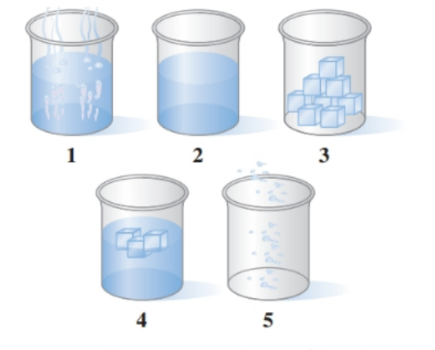
ldenify each of the following changes of state as testing. fracking, sublimation, se deposition:
a. Dry ice in an ice cream cart disappears,
h. Snowy an the ground turns to liquid water.
c. Heat is removed fromof liquids water at.
d. Frost (ice) forms of the walls of a freezer unit of a refrigerator.
What do you think about this solution?
We value your feedback to improve our textbook solutions.