Chapter 3: Q. 3.72 (page 92)
Classify each of the following as an element, a compound, or a mixture;
a. neon gas in lights
b. a salad dressing of on and vinegar
c. sodium hypochlorite (NaCIO) in bleach
Learning Materials
Features
Discover
Chapter 3: Q. 3.72 (page 92)
Classify each of the following as an element, a compound, or a mixture;
a. neon gas in lights
b. a salad dressing of on and vinegar
c. sodium hypochlorite (NaCIO) in bleach
All the tools & learning materials you need for study success - in one app.
Get started for free
Calculate each of the following temperatures in kelvins and degrees Fahrenheit:
a. The highest recorded temperature in the world was in El Azizia, Libya, on September 13, 1922.
b. The lowest recorded temperature in the world wasin Vostok, Antarctica, on July 21, 1983.
Calculate each of the following temperatures in degrees Celsius and kelvins: (3.3)
a. The highest recorded temperature in the continental United States was in Death Valley, California, on July 10,
b. The lowest recorded temperature in the continental United States was in Rodgers Pass, Montana, on January
Indicate whether each of the following describes a gas, a liquid, or a solid:
a. Lemonade has a definite volume but takes the shape of its container.
b. The particles in a tank of oxygen are very far apart.
c. Helium occupies the entire volume of a balloon.
If the same amount of heat is supplied to samples ofeach of aluminum, iron, and copper all at. which sample would reach the highest temperature (see Table 3.11)?
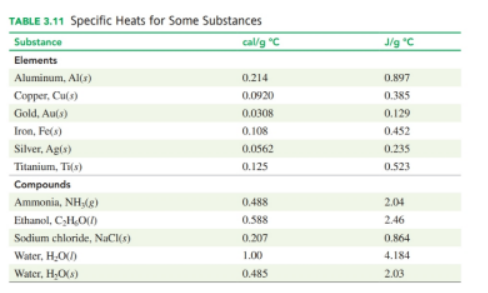
Use the heat equation to calculate the energy for each of the following (see Table 3.11 ):
a. calories to heat 8.5g of water from
b. joules lost when 25g of water cools from
c. kilocalories to heat 150g of water from
d. kilojoules to heat 175 g of copper from
What do you think about this solution?
We value your feedback to improve our textbook solutions.