Chapter 3: Q. 3.62 (page 91)
Classify each of the following as a homogeneous or heterogeneous mixture:



Short Answer
All three components are heterogeneous mixture.
Learning Materials
Features
Discover
Chapter 3: Q. 3.62 (page 91)
Classify each of the following as a homogeneous or heterogeneous mixture:



All three components are heterogeneous mixture.
All the tools & learning materials you need for study success - in one app.
Get started for free
Discuss the changes in the potential and kinetic energy of a ski jumper taking the elevator to the top of the jump and going down the ramp.
Oa a hot sunny day, you get out of the swimming pool and sit in a metal chair, which is very hot. Would you predict that the specific heat of the metal is higher or lower than that of water? Explain.
A piece of ice at is added to a sample of water at . All of the ice melts and the temperature of the water decreases to . How many grams of water were in the sample?
In a large building, oil is used in a steam boiler heating system. The combustion of of oil provides .
How many kilograms of oil are needed to heat of water from to ?
How many kilograms of oil are needed to change of water to steam at ?
Identify each of the following as an element, a compound, or a mixture. Explain your choice. (3.1)
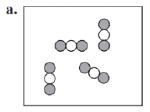
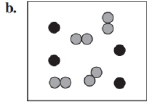
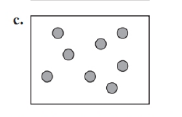
What do you think about this solution?
We value your feedback to improve our textbook solutions.