Chapter 3: Q. 3.42 (page 86)
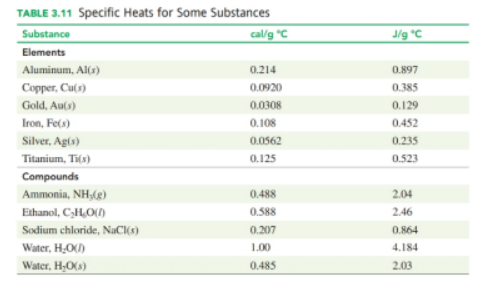
Use the heat equation to calculate the energy, in joules and calories, for each of the following (see Table 3.11):
a. to heat of water from to
b. lost when of water cools fromto
c. to heat of silver fromto
d. lost whenof gold cools fromto
Short Answer
(part a) As a result, the required heat is 311 cal.
(part b) As a result, the heat lost is
(part c) As a result, the required heat is
(part d) As a result, the heat lost is
Step by step solution
Given data (part a)
(a) The mass of water is, and the specific heat for water is localid="1653052589831" The initial temperature is is localid="1653052595971" .
The final temperature isislocalid="1653052605136" .
The heat lost can be converted from joules to calories using the conversion factor shown below.
Step 2:The tempature change (part a)
The temperature change can be calculated by using the following formula:
localid="1653052613916"
localid="1653052620116"
The temperature change is localid="1653052626606" .
The heat equation (part a)
The heat equation is as follows.
Heat mass
By substituting the value in the preceding equation, we obtain
localid="1653052638163"
localid="1653052645235"
As a result, the required heat islocalid="1653052651623"
Step 4:The heat required can be converted from joules into calories (part a)
The heat lost can be converted from joules to calories using the conversion factor shown below.
The heat required is
As a result, the required heat is .
Step 5:Given data (part b)
(b) The mass of water is , and the specific heat for water is localid="1653052717859" The initial temperature isislocalid="1653052723774" .
The final temperature isis localid="1653052729775" .
The heat lost can be converted from joules to calories using the conversion factor shown below.
Step 6:The temperature change (part b)
The temperature change can be calculated by using the following formula:
localid="1653052740468"
localid="1653052747540"
Therefore, the temperature change is localid="1653052754087" .
Step 7:The heat equation (part b)
The heat equation is as follows.
Heatmass
By substituting the value in the preceding equation, we obtain
localid="1653052765500"
The minus sign denotes that heat is lost during the process.. Therefore, the heat lost is localid="1653052771541" .
Step 8:The heat lost can be converted from joules into calories (part b)
The heat lost can be converted from joules to calories using the conversion factor shown below.:
The heat required is
Heat
As a result, the required heat is .
Step 9:Given data (part c)
(c) The mass of silver is , and the specific heat for silver islocalid="1653052830136" . The initial temperature is is localid="1653052824312" .
The final temperature is is localid="1653052838424" .
The heat lost can be converted from joules to calories using the conversion factor shown below.
Step 10:The temperature change (part c)
The temperature change can be calculated by using the following formula:
localid="1653052848805"
localid="1653052855023"
As a result, the required heat is localid="1653052861088" .
Step 11:The heat equation (part c)
The heat equation is as follows.
Heatmass
By substituting the value in the preceding equation, we obtain
Heat localid="1653052871100"
localid="1653052877473"
As a result, the required heat is localid="1653052883588"
Step 12:The heat required can be converted from joules into calories (part c)
The heat lost can be converted from joules to calories using the conversion factor shown below.
The heat required is
As a result, the required heat is .
Step 13:Given data (part d)
(d) The mass of gold is , and the specific heat)for iron is localid="1653052968300" . The initial temperature is islocalid="1653052974647" .
The final temperature isis localid="1653052981118" .
We must calculate the required heat in joules and calories.
Step 14:The temperature change (part d)
The temperature change can be calculated by using the following formula:
localid="1653052992535"
localid="1653052999068"
Therefore, the temperature change is localid="1653053006237"
The heat equation is
Heatmass
By substituting the value in the preceding equation, we obtain
localid="1653053015039"
localid="1653053025035"
The minus sign indicates that the heat is lost in the process. Therefore, the heat lost is.
The heat lost can be converted from joules to calories using the conversion factor shown below
localid="1653053054078"
localid="1653053062424"
The heat lost is
Heat localid="1653053073495"
localid="1653053087241"
= 588 cal
As a result, the required heat is 58.8 cal.

Unlock Step-by-Step Solutions & Ace Your Exams!
-
Full Textbook Solutions
Get detailed explanations and key concepts
-
Unlimited Al creation
Al flashcards, explanations, exams and more...
-
Ads-free access
To over 500 millions flashcards
-
Money-back guarantee
We refund you if you fail your exam.
Over 30 million students worldwide already upgrade their learning with 91Ӱ��!