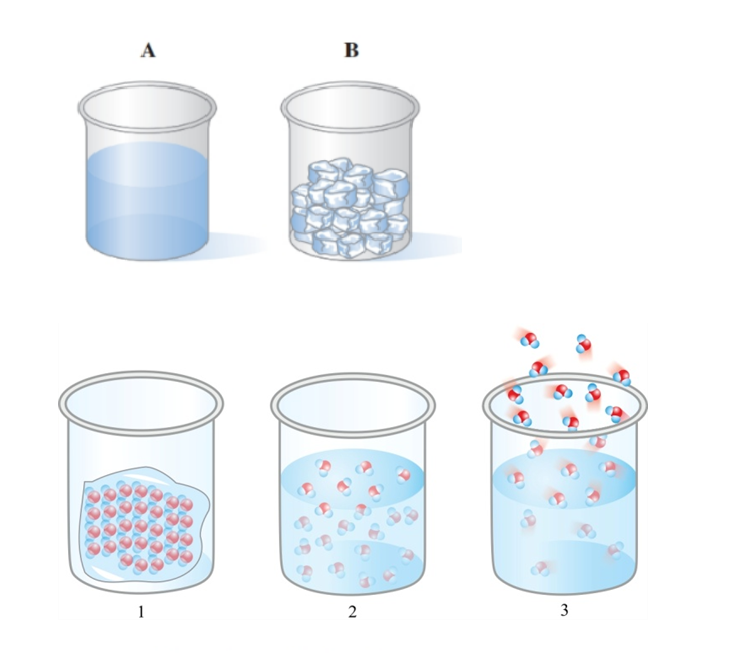
Answer the following for the water samples and shown in the diagram
In which sample (or ) does the water have its own shape?
Which diagram ( or or ) represents the arrangement of particles in water sample ?
Which diagram ( or or ) represents the arrangement of particles in water sample ?
Answer the following for diagrams :
The state of matter indicated in diagramis a_____ ; in diagram , it is a ____; and in diagram , it is a____.
The motion of the particles is slowest in diagram_____.
The arrangement of particles is farthest apart in diagram_______.
The particles fill the volume of the container in diagram_______.
If the water in diagram has a mass of and a temperature of , how much heat, in kilojoules, is removed to cool the liquid to?![]()