Chapter 15: Q.15.85 (page 542)
Draw the condensed structural formula for a glycerophospholipid that contains glycerol, two stearic acids, phosphate, and ethanolamine (ionized).
Short Answer
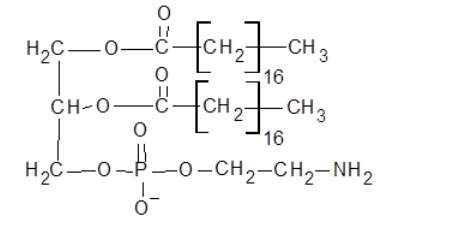
The condensed structural formula is,
Learning Materials
Features
Discover
Chapter 15: Q.15.85 (page 542)
Draw the condensed structural formula for a glycerophospholipid that contains glycerol, two stearic acids, phosphate, and ethanolamine (ionized).
The condensed structural formula is,

All the tools & learning materials you need for study success - in one app.
Get started for free
Identify each of the following processes as hydrogenation, hydrolysis, or saponification and give the products:
a. the reaction of palm oil with
b. the reaction of glyceryl trilinoleate from safflower oil with water and .
Use condensed structural formulas to write the balanced chemical equation for the hydrogenation of glyceryl tripalmitoleate, a fat containing glycerol and three palmitoleic acid molecules.
Which of the following solvents might be used to dissolve an oil stain?
a. water
b.
c. diethyl ether
d. benzene
e. solution
Olive oil is monounsaturated, whereas hutter fot is caturated. Why does olive oil have a lower meltiag point than butser far?
Use condensed structural formulas to write the balanced chemical equation for the saponification of glyceryl trioleate (triolein).
What do you think about this solution?
We value your feedback to improve our textbook solutions.