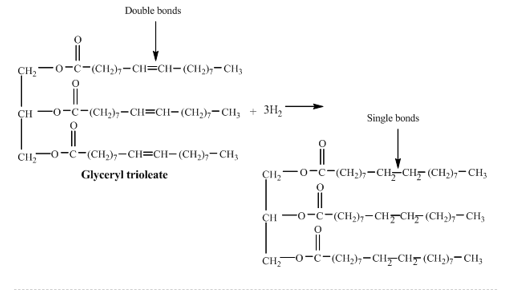
Chapter 15: Q. 15.91 (page 543)
mole of glyceryl trioleate (triolein) is completely hydrogenated.
a. Draw the condensed structural formula for the product.
b. How many moles of hydrogen are required?
c. How many grams of hydrogen are required?
d. How many liters of hydrogen gas are needed if the reaction is run at STP?
Short Answer
(Part a)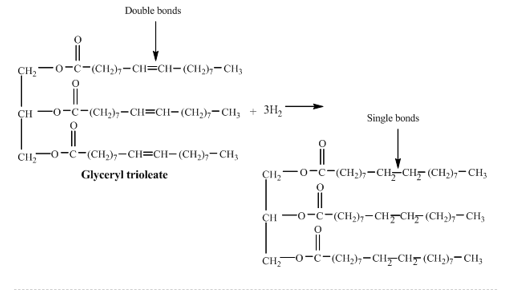
(Part b) Thorough halogenation of lecithin crystallisation takes three equivalents of protons.
(Part c) Number of grams of biogas required for a completed dehydrogenation process .
(Part d) of hydrogen gas required for full hydrogenation reaction.