Chapter 11: Q. 11.61 (page 394)
Write a chemical equation for complete combustion for each of the following:
a)dimethylpropane
b) cyclobutane
c)hexane
Short Answer
The chemical equations for complete combustion are:
a)
b)
c)
Learning Materials
Features
Discover
Chapter 11: Q. 11.61 (page 394)
Write a chemical equation for complete combustion for each of the following:
a)dimethylpropane
b) cyclobutane
c)hexane
The chemical equations for complete combustion are:
a)
b)
c)
All the tools & learning materials you need for study success - in one app.
Get started for free
Name each of the following aromatic compounds:

Convert each of the following line-angle formulas to a condensed structural formula and give its IUPAC name: ()
a.
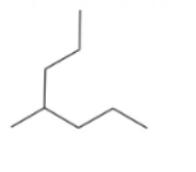
b.
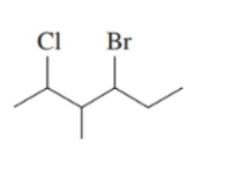
Write the balanced chemical equation for the complete combustion of each of the following compounds:
a. hexane
b. cyclopentane
c. dimethylpropane
Give the name of the product from the hydrogenation of each of the following:
a)hexene
b) methylpentene
c) cyclopropene
Draw the condensed structural formulas for all the possible haloalkane isomers that have four carbon atoms and a bromine.
What do you think about this solution?
We value your feedback to improve our textbook solutions.