Chapter 8: Q. 8.56 (page 278)
In a gas mixture, the partial pressure are argon , neon , and nitrogen . What is the total pressure, in , exerted by the gas mixture?
Short Answer
The total pressure is.
Learning Materials
Features
Discover
Chapter 8: Q. 8.56 (page 278)
In a gas mixture, the partial pressure are argon , neon , and nitrogen . What is the total pressure, in , exerted by the gas mixture?
The total pressure is.
All the tools & learning materials you need for study success - in one app.
Get started for free
Use the kinetic molecular theory of gases to explain each of the following:
At which of the following diagrams represents a gas sample that exerts the:
a. lowest pressure?
b. highest pressure?
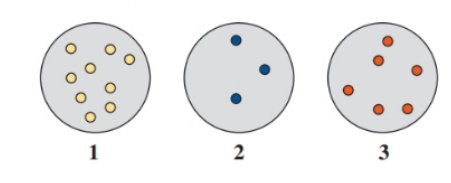
Why do scuba divers need to exhale air when they ascend to the surface of the water?
Use the words inspiration and expiration to describe the part of the breathing cycle that occurs as a result of each of the following:
a. The diaphragm contracts.
b. The volume of the lungs decreases.
c. The pressure within the lungs is less than that of the atmosphere.
Sometimes when you blow up a balloon and release it, it flies around the room. What is happening to the air in the balloon and its volume?
What do you think about this solution?
We value your feedback to improve our textbook solutions.