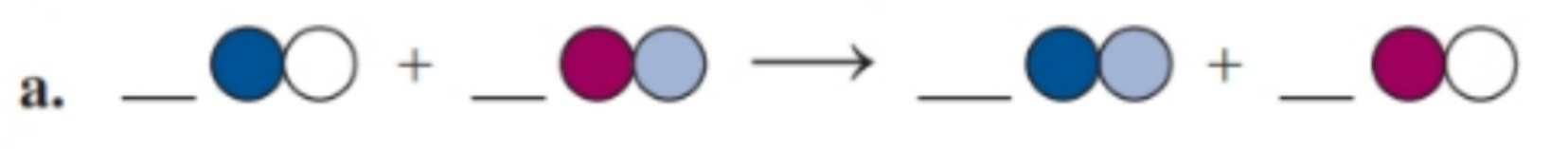Chapter 7: Q. 7.84 (page 254)
Balance each of the following by adding coefficients, and identify the type of reaction for each
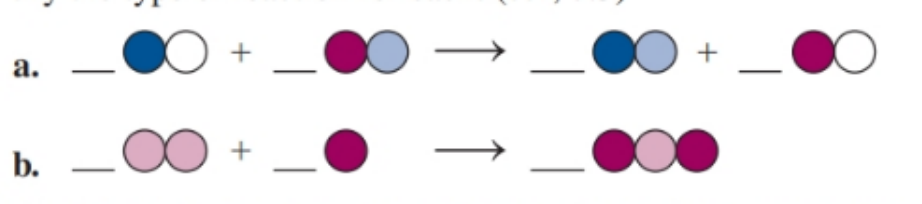
Short Answer
(a) The balanced chemical equation is Substitution reaction.
(b) The balanced chemical equation is Addition reaction.