Chapter 7: Q. 7.13 (page 223)
Calculate the molar mass for each of the following :
a. b. c.
Short Answer
a. The molar mass of islocalid="1653486070991"
b. The molar mass of islocalid="1653486060297"
c. The molar mass of islocalid="1653486050502"
Learning Materials
Features
Discover
Chapter 7: Q. 7.13 (page 223)
Calculate the molar mass for each of the following :
a. b. c.
a. The molar mass of islocalid="1653486070991"
b. The molar mass of islocalid="1653486060297"
c. The molar mass of islocalid="1653486050502"
All the tools & learning materials you need for study success - in one app.
Get started for free
Identify each of the following as an oxidation or a reduction:
a.
b.
c.
d.
a. How does a catalyst affect the activation energy?
b. Why is pure oxygen used in respiratory distress?
Balance each of the following by adding coefficients, and identify the type of reaction for each:
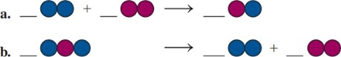
Calculate the mass, in grams, for each of the following:
a.moles of
b. moles of
c. mole of
d. mole of
e. moles of
In the mitochondria of human cells, energy is provided by the oxidation and reduction reactions of the iron ions in the cytochromes in electron transport. Identify each of the following as an oxidation or a reduction:
a.
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.