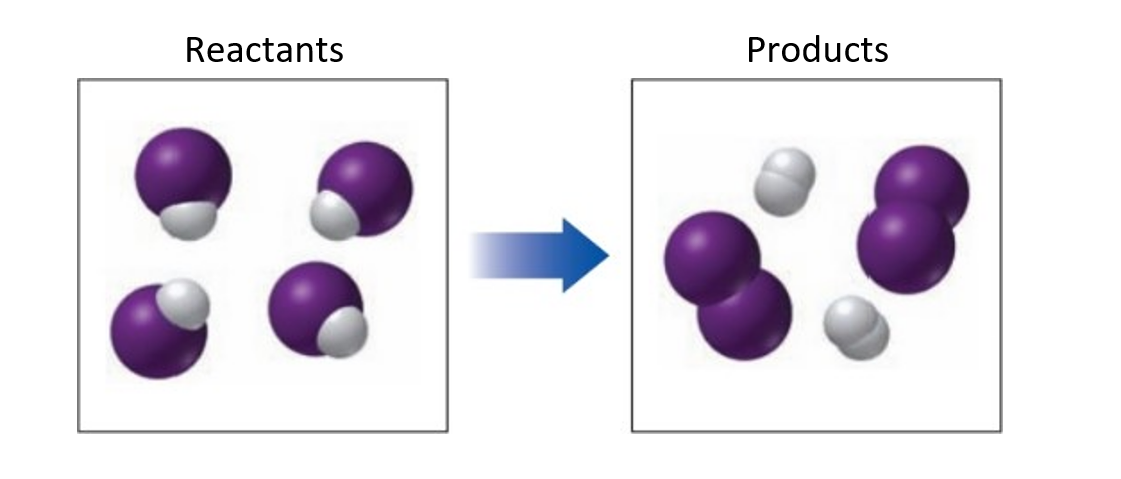
Chapter 7: Q. 7.42 (page 237)
Classify each of the following as a combination, decomposition, single replacement, double replacement, or combustion reaction:
a.
b.
c.
d.
e.
Short Answer
(a) The reaction is combination reaction.
(b) The reaction is double displacement reaction.
(c) The reaction is decomposition reaction.
(d) The reaction is displacement reaction.
(e) The reaction is combustion reaction.