Chapter 7: Q. 7.2 (page 220)
What is Avogadro's number
Short Answer
Avogadro's number, which is equal to , is the number of units in one mole of any substance.
Learning Materials
Features
Discover
Chapter 7: Q. 7.2 (page 220)
What is Avogadro's number
Avogadro's number, which is equal to , is the number of units in one mole of any substance.
All the tools & learning materials you need for study success - in one app.
Get started for free
Balance each of the following by adding coefficients, and identify the type of reaction for each
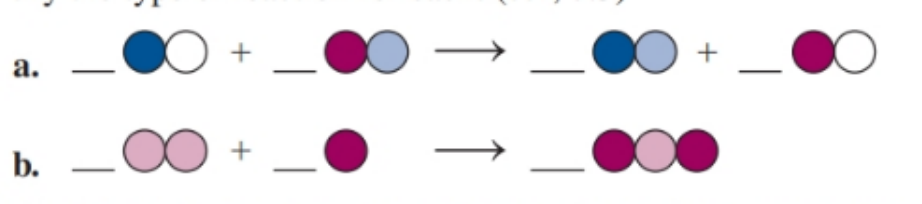
Gasohol is a fuel containing liquid ethanol () that burns in oxygen gas to give carbon dioxide and water gases. ()
a. Write the balanced chemical equation.
b. How many moles of are needed to completely react with localid="1653474292769">
c. If a car produces localid="1653474309450">
d. If you burn localid="1653475324715" gof , how many grams of and can be produced?
Identify each of the following as an oxidation or a reduction:
a.
b.
c.
d.
Consider the following unbalanced equation: ()
a. Write the balanced chemical equation.
b. Identify the type of reaction.
c. How many moles of are needed to react with localid="1653475915594" mole of ?
d. How many grams of are produced when localid="1653475936440" gof reacts?
e. When is reacted with localid="1653475950535" gof , how many grams of can form?
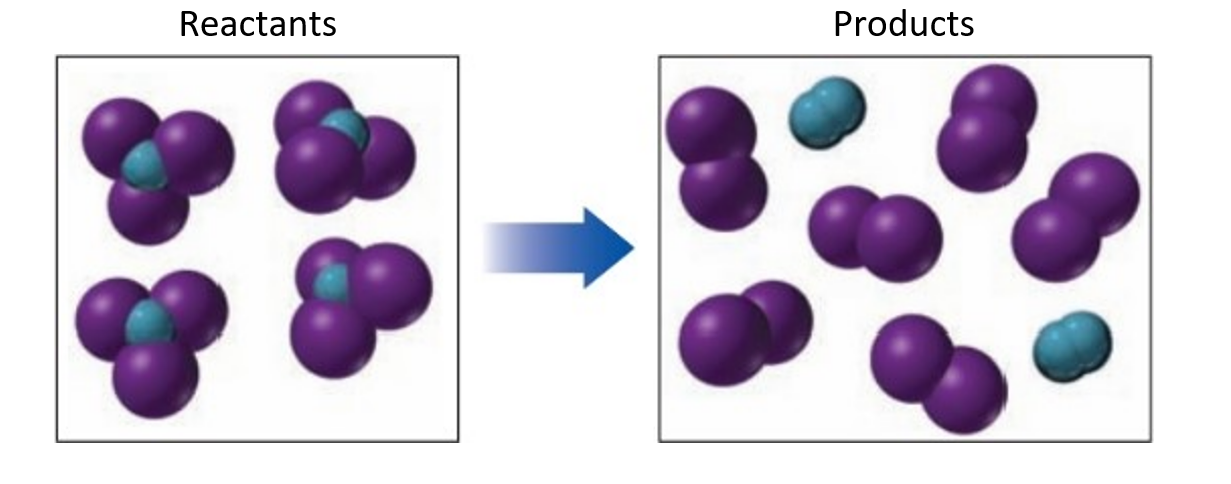
If blue spheres represent nitrogen atoms, purple spheres represent iodine atoms and the reacting molecules are solid, while the products are gases.
What do you think about this solution?
We value your feedback to improve our textbook solutions.