Chapter 14: Q. 14.36 (page 491)
Indicate if each of the following is soluble in water. Explain.
a. 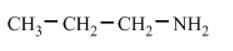
b. 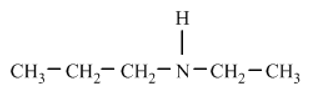
c. 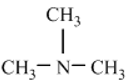
d.
Short Answer
(part a) Water soluble.
(part b) Water soluble.
(part c) Water soluble.
(part d) Water insoluble.
Learning Materials
Features
Discover
Chapter 14: Q. 14.36 (page 491)
Indicate if each of the following is soluble in water. Explain.
a. 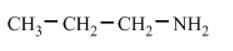
b. 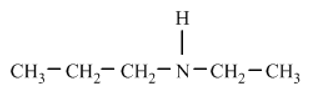
c. 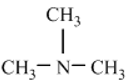
d.
(part a) Water soluble.
(part b) Water soluble.
(part c) Water soluble.
(part d) Water insoluble.
All the tools & learning materials you need for study success - in one app.
Get started for free
Write the common name and classify each of the following compounds as primary , secondary , or tertiary
a)
b)
c)
Write the IUPAC and common names, if any, for each of the following:
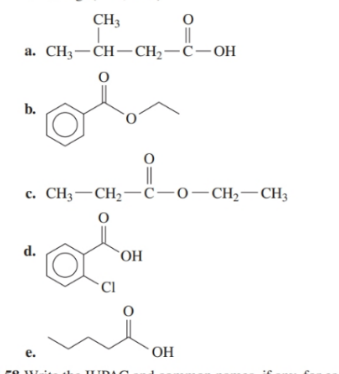
14.13 Write the IUPAC and common names, if any, of the carboxylate salts produced in problem
Draw the condensed structural formula, or line-angle formula if cyclic, for each of the following amines:
a. ethylamine
b. -methylaniline
c. butyl propylamine
Draw the condensed structural formula for each of the following amides:
a. heptolamide
b. chlorobenzamide
c. methyl butyramide
d. ethylpropanamide
What do you think about this solution?
We value your feedback to improve our textbook solutions.