Chapter 4: Q. 4.65 (page 126)
Fill in the following blanks using larger or smaller, more metallic or less metallic. Na has a atomic size and is than P.
Short Answer
Na has a larger atomic size and is more metallic than P.
Learning Materials
Features
Discover
Chapter 4: Q. 4.65 (page 126)
Fill in the following blanks using larger or smaller, more metallic or less metallic. Na has a atomic size and is than P.
Na has a larger atomic size and is more metallic than P.
All the tools & learning materials you need for study success - in one app.
Get started for free
For each representation of a nucleus through, write the atomic symbol and identify which are isotopes.
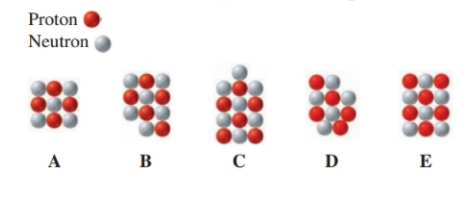
Two isotopes of rubidium occur naturally, and. Which isotope of rubidium is more abundant?
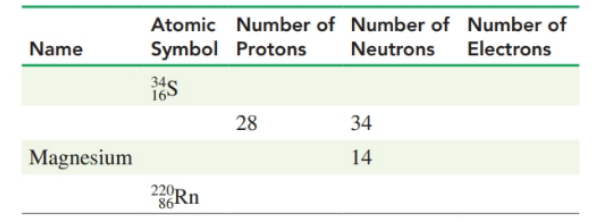
Complete the following table:
Write the names for the elements in each of the following formulas of compounds used in medicine :
a. table salt,
b. plaster casts,
c. Demerol,
d. treatment of bipolar disorder,
Place the elements in each set in order of decreasing atomic size.
a. Al, Si, Mg
b. Cl, I, Br
c. Sb, Sr, I
d. P, Si, Na
What do you think about this solution?
We value your feedback to improve our textbook solutions.