Chapter 4: Q. 4.101 (page 132)
Complete the following table:
Short Answer
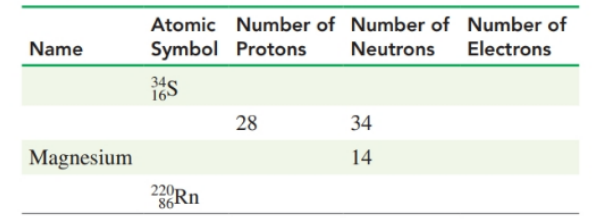
| Name | Atomic Symbol | Number of Protons | Number of Neutrons | Number of Electrons |
| Selenium | 16 | 18 | 16 | |
| Nikal | 28 | 34 | 28 | |
| Magnesium | 12 | 14 | 12 | |
| Radium | 88 | 140 | 88 |
Learning Materials
Features
Discover
Chapter 4: Q. 4.101 (page 132)
Complete the following table:

| Name | Atomic Symbol | Number of Protons | Number of Neutrons | Number of Electrons |
| Selenium | 16 | 18 | 16 | |
| Nikal | 28 | 34 | 28 | |
| Magnesium | 12 | 14 | 12 | |
| Radium | 88 | 140 | 88 |
All the tools & learning materials you need for study success - in one app.
Get started for free
Place the following in order of increasing metallic character: Na, P, AI, Ar.
The most abundant isotope of lead is .
a. How many protons, neutrons, and electrons are in ?
b. What is the atomic symbol of another isotope of lead with neutrons?
c. What is the atomic symbol and name of an atom with the same mass number as in part b and neutrons?
How did Thomson determine that the electrons have a negative charge?
Write the group number and draw the Lewis symbol for each of the following elements:
a. carbon
b. oxygen
c. argon
d. lithium
e. chlorine
What is the group number and number of valence electrons for each of the following elements?
a. Magnesium
b. Iodine
c. Oxygen
d. Phosphorus
e. Tin
f. Boron
What do you think about this solution?
We value your feedback to improve our textbook solutions.