Chapter 16: Q. 16.52 (page 576)
What is the molar mass of hydroxyurea?
Short Answer
The molar mass of hydroxyurea is
Learning Materials
Features
Discover
Chapter 16: Q. 16.52 (page 576)
What is the molar mass of hydroxyurea?
The molar mass of hydroxyurea is
All the tools & learning materials you need for study success - in one app.
Get started for free
Seeds and vegetables are often deficient in one or more essential amino acids. Using the table in problem, state whether each combination provides all of the essential amino acids.

a. rice and lima beans.
b. rice and oatmeal.
c. oatmeal and lima beans.
Identify the amino acids and type of interaction that occurs between the following R groups in tertiary protein structures:
a)

b)

c)

d)
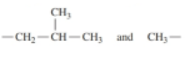
16.12 Draw the condensed structural formula for cach of the following peptides, and give its three-letler and one-letler abbreviations:
a. prolyl aspartate
b. threonyl leucine
c. methionylglutaminyllysine
d. histidylglycylghutamylisoleucine
If glutamate were replaced by proline in a protein, how might the tertiary structure be affected?
If Jeremy's current weight is , how many milligrams of hydroxyurea should Jeremy be given each day?
What do you think about this solution?
We value your feedback to improve our textbook solutions.