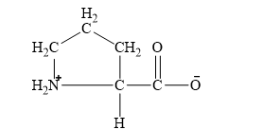
Chapter 16: Q. 16.67 (page 581)
If glutamate were replaced by proline in a protein, how might the tertiary structure be affected?
Short Answer
The replacement of the protein glutamate with proline affects tertiary structure, the reasons are
- Within a protein, amino acids with non-polar groups gather far from the aqueous environment to form a hydrophobic center within the protein molecule.