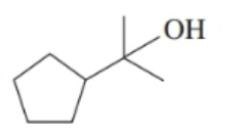
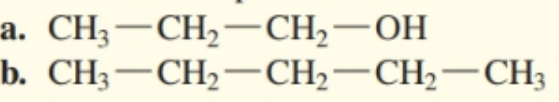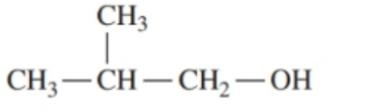Chapter 12: Q. 12.31 (page 420)
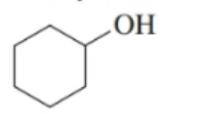
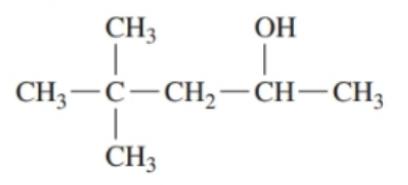
Draw the condensed structural formulas for the aldehyde and carboxylic acid produced when each of the following is oxidized:
Short Answer
a. first oxidized to and it further oxidized to .
b. first oxidizes to and then to .
c. first oxidizes to and further oxidizes to