Chapter 12: Q. 12.12 (page 408)
Are each of following alcohol is soluble, slightly soluble or insoluble in water.
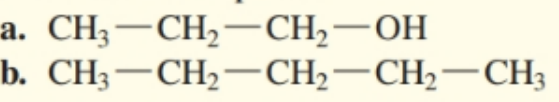
Short Answer
(a) Given compound is soluble in water .
(b) Given compound is insoluble in water .
Learning Materials
Features
Discover
Chapter 12: Q. 12.12 (page 408)
Are each of following alcohol is soluble, slightly soluble or insoluble in water.
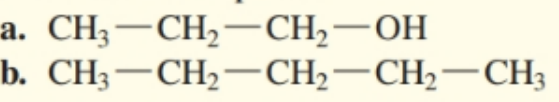
(a) Given compound is soluble in water .
(b) Given compound is insoluble in water .
All the tools & learning materials you need for study success - in one app.
Get started for free
Identify each of the compounds as aldehyde or ketone.
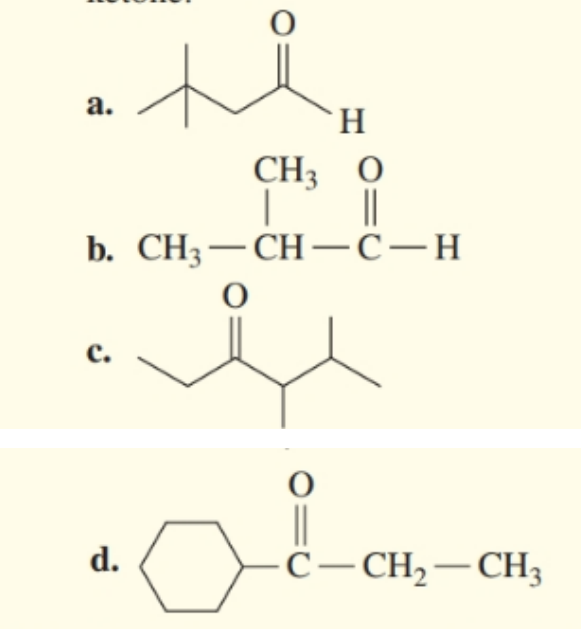
Draw the condensed structural formula for each of the following :
(a)
(b)
(c)
(d)
Which of the following will give a positive Tollens' test?
a. propanal
b. ethanol
c. ethyl methyl ether
Draw the condensed structural or line-angle formula for the alcohol produced when hydrogen and a nickel catalyst reduce each of the following: ()
a.
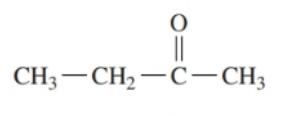
b.
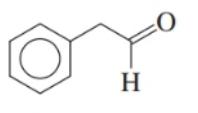
c.
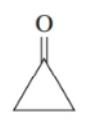
Draw the condensed structure or line formula of each of following:
(a) Formaldehyde
(b) chloro butanal
(c) methyl role="math" localid="1653538534568" hexanone
(d) dimethyl hexanal
What do you think about this solution?
We value your feedback to improve our textbook solutions.