Chapter 12: Q. 12.19 (page 413)
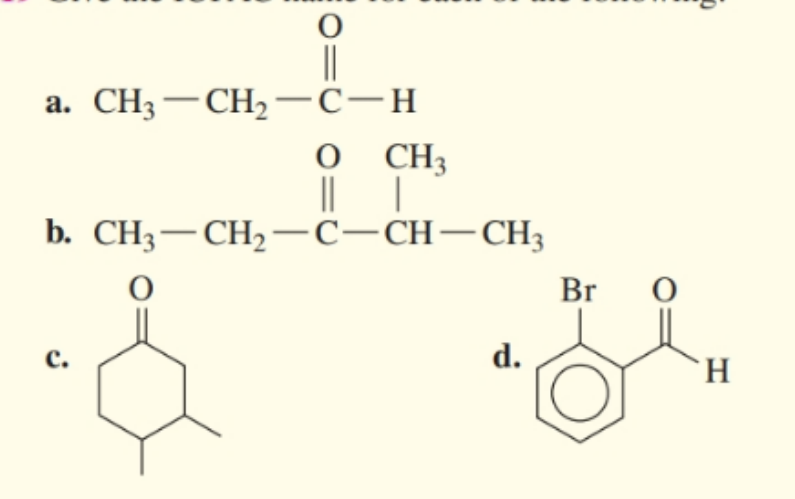
Give IUPAC name for each of the following.
Short Answer
(a) IUPAC name is Propenal .
(b) IUPAC name is .
(c) IUPAC name is .
(d) IUPAC name is localid="1652762698568" .
Learning Materials
Features
Discover
Chapter 12: Q. 12.19 (page 413)
Give IUPAC name for each of the following.

(a) IUPAC name is Propenal .
(b) IUPAC name is .
(c) IUPAC name is .
(d) IUPAC name is localid="1652762698568" .
All the tools & learning materials you need for study success - in one app.
Get started for free
Which compound in each of the following pairs would be more soluble in water explain.
a.
 or
or
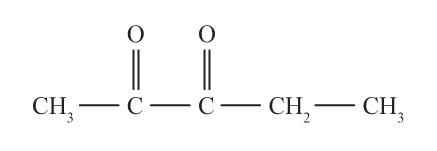
b. acetone or 2-pentanone
c. propanal or pentanal
One of the components of gasoline is octane, , which has a density of . The combustion of mole of octane provides . A hybrid car has a fuel tank with a capacity of and a gas mileage of
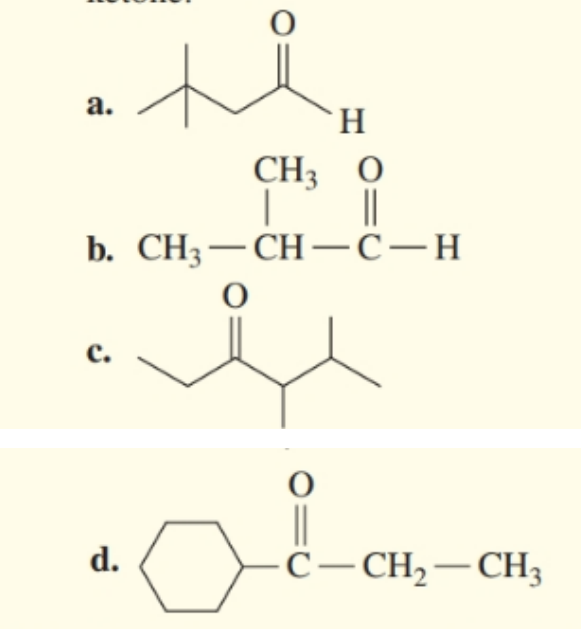
Identify each of the compounds as aldehyde or ketone.
Draw the condensed structural formula, or line-angle formula if cyclic, for each of the following:
a. propyl alcohol
b. 3-pentanethiol
c.2-methyl -2-butanol
d. 4-bromophenol
Draw the condensed structural or line-angle formula for the aldehyde or ketone formed when each of the following alcohols is oxidized [O] (if no reaction, write none):
What do you think about this solution?
We value your feedback to improve our textbook solutions.