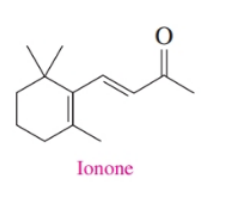
Ionone is a compound that gives violets their aroma. The small, edible, purple flowers of violets are used in salads and to make teas. Liquid ionone has a density of
a. What functional groups are present in ionone?
b. Is the double bond on the side chain cis or trans?
c. What are the molecular formula and molar mass of ionone?
d. How many moles are in of ionone ?
e. When ionone reacts with hydrogen in the presence of a platinum catalyst, hydrogen adds to the double bonds and converts the ketone group to an alcohol. Draw the condensed structural formula and give the molecular formula for the product.
f. How many milliliters of hydrogen gas at STP are needed to completely react with of ionone?
 or
or
 is more soluble.
is more soluble. and
and
 is more soluble due to hydrogen bonding. Hydrogen bonding is the interaction that involves a hydrogen atom and pair of other atoms.
is more soluble due to hydrogen bonding. Hydrogen bonding is the interaction that involves a hydrogen atom and pair of other atoms. 


