Chapter 10: Q. 10.48 (page 350)
Balance each of the following neutralization equations:
a.
b.localid="1654005825158"
Short Answer
The neutralization equation is balanced
a.
b.
Learning Materials
Features
Discover
Chapter 10: Q. 10.48 (page 350)
Balance each of the following neutralization equations:
a.
b.localid="1654005825158"
The neutralization equation is balanced
a.
b.
All the tools & learning materials you need for study success - in one app.
Get started for free
Solutionhas a pH of, and solution B has aof. (10.6)
a. Which solution is mone acidic?
b. What is thelocalid="1653977618353" in each?
c. What is the localid="1653977632191" in each?
If of a solution is required to titrate of a solution of , what is the molarity of the solution
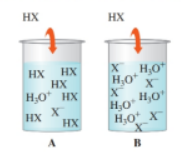
Determine if each of the following diagrams represents a strong acid or a weak acid. The acid has the formula HX. (10.3)
Use Le Châtelier's principle to predict if each of the following changes causes the system to shift in the direction of products or reactants:
a. adding more
b. removing some
c. adding more
d. removing some
Calculate the volume, in milliliters, of a solution that will complesely neutralize each of the following: (10.7)
a. of a ().286 M HC7 solution
b. of a solution
c.of a HBr solution
What do you think about this solution?
We value your feedback to improve our textbook solutions.