Chapter 12: Q2. (page 398)
The hypothetical elementary reaction has a rate constant of . What is the reaction velocity when the concentration of is ?
Short Answer
The reaction velocity for the hypothetical elementary reaction is 10-10Ms-1.
Learning Materials
Features
Discover
Chapter 12: Q2. (page 398)
The hypothetical elementary reaction has a rate constant of . What is the reaction velocity when the concentration of is ?
The reaction velocity for the hypothetical elementary reaction is 10-10Ms-1.
All the tools & learning materials you need for study success - in one app.
Get started for free
Why are such allosteric enzymes composed of more than one catalytic subunit?
The KM for the reaction chymotrypsin with N-acetylvaline ethyl ester is , and the for the reaction of chymotrypsin with N-acetyltyrosine ethyl ester is . (a) Which substrate has the higher apparent affinity for the enzyme? (b) Which substrate is likely to give a higher value for ?
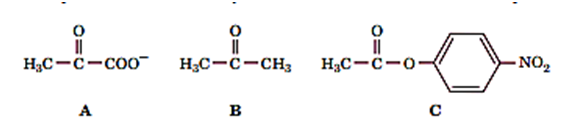
Molecule A is the substrate for enzyme X. Which is more likely to be a competitive inhibitor of enzyme X: molecule B or molecule C? Explain.
From the reaction data below, determine whether the reaction is first order or second order and calculate the rate constant.
Time (s) | Reactant (mM) |
0 | 5.4 |
1 | 4.6 |
2 | 3.9 |
3 | 3.2 |
4 | 2.7 |
5 | 2.3 |
For an enzymatic reaction, draw curves that show the appropriate relationships between the variables in each plot below.
What do you think about this solution?
We value your feedback to improve our textbook solutions.