Chapter 12: Q1. (page 373)
Write the rate equations for a first-order and a second-order reaction.
Short Answer
The rate equation for a first-order reaction is and the rate equation for a second-order reaction is .
Learning Materials
Features
Discover
Chapter 12: Q1. (page 373)
Write the rate equations for a first-order and a second-order reaction.
The rate equation for a first-order reaction is and the rate equation for a second-order reaction is .
All the tools & learning materials you need for study success - in one app.
Get started for free
Derive the Michaelis–Menten equation.
From the reaction data below, determine whether the reaction is first order or second order and calculate the rate constant.
Time | Reactant |
0 | 6.2 |
1 | 3.1 |
2 | 2.1 |
3 | 1.1 |
4 | 1.3 |
| 5 | 1.1 |
How does cytochrome P450 participate in drug metabolism?
You are constructing a velocity versus [substrate] curve for an enzyme whose KM is believed to be about 2 μM. The enzyme concentration is 200 nM and the substrate concentrations range from 0.1 μM to 10 μM. What is wrong with this experimental setup and how could you fix it?
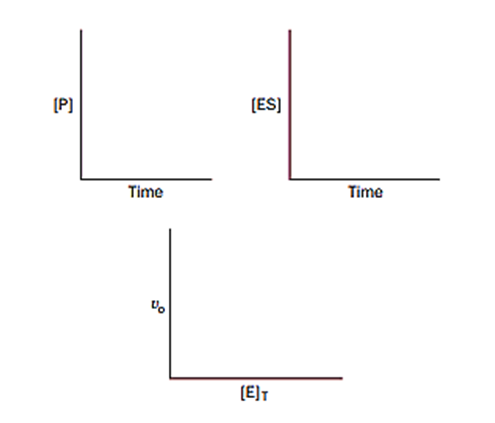
For an enzymatic reaction, draw curves that show the appropriate relationships between the variables in each plot below.
What do you think about this solution?
We value your feedback to improve our textbook solutions.