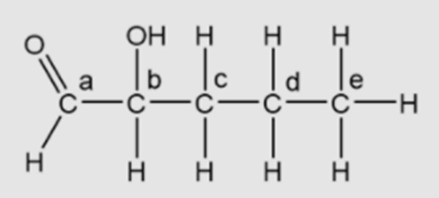
Chapter 4: Q7TYU (page 56)
Which action could produce a carbonyl group?
(A) the replacement of the –OH of a carboxyl group with hydrogen
(B) the addition of a thiol to a hydroxyl
(C) the addition of a hydroxyl to a phosphate
(D) the replacement of the nitrogen of an amine with oxygen
Short Answer
(A) The option “the replacement of the –OH of a carboxyl group with hydrogen” is true.
(B) The option “the addition of a thiol to a hydroxyl” is false.
(C) The option “the addition of a hydroxyl to a phosphate” is false.
(D) The option “the replacement of the nitrogen of an amine with oxygen” is false.