Chapter 4: Q6TYU (page 56)
Identify the asymmetric carbon in this molecule:
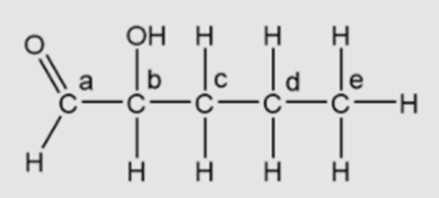
Short Answer
(a) The option “a” is false.
(b) The option “b” is true.
(c) The option “c” is false.
(d) The option “d” is false.
(e) The option “e” is false.
Learning Materials
Features
Discover
Chapter 4: Q6TYU (page 56)
Identify the asymmetric carbon in this molecule:

(a) The option “a” is false.
(b) The option “b” is true.
(c) The option “c” is false.
(d) The option “d” is false.
(e) The option “e” is false.
All the tools & learning materials you need for study success - in one app.
Get started for free
Why was Wohler astonished to find he had made urea?
Which action could produce a carbonyl group?
(A) the replacement of the –OH of a carboxyl group with hydrogen
(B) the addition of a thiol to a hydroxyl
(C) the addition of a hydroxyl to a phosphate
(D) the replacement of the nitrogen of an amine with oxygen
Suppose you had an organic molecule such as cysteine (see Figure 4.9, sulfhydryl group example), and you chemically removed the —NH2 group and replaced it with —COOH. Draw this structure. How would this change the chemical properties of the molecule? Is the central carbon asymmetric before the change? After?
Which chemical group is most likely to be responsible for an organic molecule behaving as a base (see Concept 3.3)
hydroxyl
carbonyl
amino
phosphate
The synthesis of products is limited by the amount of reactants. (a) If one mole each of CH4, NH3, H2S, and CO2 is added to \({\bf{1}}\) liter of water in a flask, how many moles of hydrogen, carbon, oxygen, nitrogen, and sulfur are in the flask?
(b) Looking at the molecular formula in the table, how many moles of each element would be needed to make \({\bf{1}}.{\bf{0}}\) mole of glycine?
(c) What is the maximum number of moles of glycine that could be made in that flask, with the specified ingredients, if no other molecules were made? Explain.
(d) If serine or methionine were made individually, which element(s) would be used up first for each? How much of each product could be made?
What do you think about this solution?
We value your feedback to improve our textbook solutions.