Chapter 3: Q60P (page 644)
You compress a gas in an insulated cylinder— no heat flows into or out of the gas. The gas pressure is fairly low, so treating the gas as ideal is a good approximation. When you measure the pressure as a function of the volume of the gas, you obtain these results:

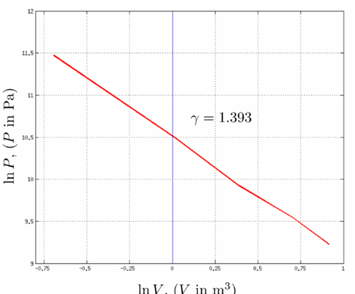
(a) Graph log 1p2 versus log 1V2, with p in Pa and V in m3 . Explain why the data points fall close to a straight line. (b) Use your graph to calculate g for the gas. Is the gas monatomic, diatomic, or polyatomic? (c) When p = 0.101 atm and V = 2.50 L, the temperature is 22.0o C. Apply the ideal-gas equation and calculate the temperature for each of the other pairs of p and V values. In this compression, does the temperature of the gas increase, decrease, or stay constant?
Short Answer
(a) comes with negative slope so the data points fall close to a straight line.
(b)the gas is monoatomic.
(c) T(22.0oC) < T2(55.056oC) < T3(76.306oC) < T4(153.05oC) < T5(283.25oC)
temperature of gas increases as pressure increases.