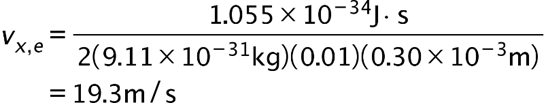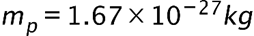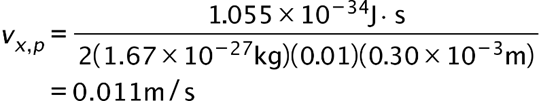Chapter 6: Q48E (page 1316)
(a) The x-coordinate of an electron is measured with an uncertainty of 0.30 mm. What is the x-component of the electron’s velocity, if the minimum percent uncertainty in a simultaneous measurement of
if the minimum percent uncertainty in a simultaneous measurement of is 1.0% ?(b) Repeat part (a) for a proton.
is 1.0% ?(b) Repeat part (a) for a proton.
Short Answer
(a) The x-component of the electron’s velocity is 19.3m/s.
(b) The x-component of the proton’s velocity is 0.011m/s.
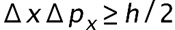
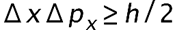
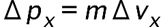
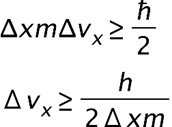
 is the uncertainty on the x-component.
is the uncertainty on the x-component. is the uncertainty in an electron's x-coordinate,
is the uncertainty in an electron's x-coordinate, is the mass of the electron, and
is the mass of the electron, and is the minimum percent uncertainty in a simultaneous measurement of speed.
is the minimum percent uncertainty in a simultaneous measurement of speed.